Distinction between obstructive and non-obstructive azoospermia
Prior to initiating treatment for a couple, in whom the man has azoospermia, it is important to distinguish whether the lack of sperm in the ejaculate is from an obstructive or non-obstructive process. In addition, careful evaluation of the post-ejaculate urinalysis is necessary to rule out retrograde ejaculation, common in diabetics. Men with retrograde ejaculation do not require invasive procedures for sperm retrieval; sperm may be isolated from urine or catheterized from the bladder and used for intrauterine insemination or IVF. Paraplegics may be treated with vibratory stimulation or electro-ejaculation and do not require surgical sperm retrieval. History and physical examination can suggest the cause of azoospermia for an experienced examiner. For example, the presence of normal volume testes with bilaterally indurated epididymides and/or absent vas deferens will point to an obstructive etiology for azoospermia. A history of cryptorchidism in the presence of small or soft testes suggests non-obstructive azoospermia (NOA), especially if associated with an elevated serum (FSH) level.
The distinction between obstructive and non-obstructive azoospermia is important. Men with obstructive azoospermia may have other cost-effective options for treatment, such as microsurgical reconstruction of the reproductive tract. Also, a real risk of failure to retrieve spermatozoa exists in men with non-obstructive azoospermia (NOA), and couples must be apprised of this risk before attempting assisted reproduction with sperm retrieval and ICSI. Men with NOA azoospermia often have a genetic abnormality that has caused the lack of sperm, whereas most men with congenital absence of the vas deferens have abnormalities of the cystic fibrosis (CFTR) gene.
Sperm quality in obstructive and non-obstructive azoospermia
In the normal male reproductive tract, sperm exiting the testis have minimal motility and limited egg fertilizing capacity. Sperm acquire the potential for improved motility and fertilizing ability during epididymal transit. So, in the unobstructed epididymis, sperm of optimal quality (as evaluated by percent motile cells) are found in the most distal epididymis.
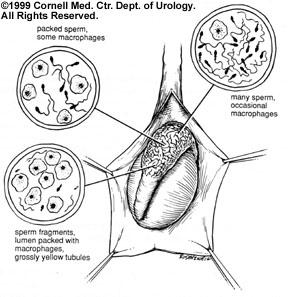
1. Obstructive Azoospermia: The obstructed epididymis or congenital absence of the vas deferens shows the opposite pattern of sperm quality: optimal sperm quality in the proximal epididymis and very poor quality in the most distal segments. This finding of "inverted motility" is expected in the obstructed male reproductive tract since sperm production that continues in the testis and reabsorption of those sperm is an active process in the most distal regions of the system. The most distal obstructed epididymis tends to contain dilated yellow tubules that are packed with macrophages reabsorbing old, degenerated sperm.9 Therefore, sperm retrieval should be performed from the proximal obstructed epididymis and testis, and higher quantities of motile spermatozoa can be obtained. In fact, motile spermatozoa are found at concentrations up to 1 million sperm per microliter in the fluid of obstructed epididymis.
2. Non-obstructive Azoospermia: A common observation for testicular sperm samples is that retrieved spermatozoa are immobile or have a sluggish twitching motion. In this situation, some sperm production is focally present within the testis, despite the fact that inadequate numbers of sperm are released from the testis to make it into the ejaculate. After several hours of incubation in vitro, testicular sperm typically show some motility. The lack of initial motility does not necessarily reflect a lack of viability for testicular sperm, since these sperm have never acquired motility. Non-motile ejaculated sperm have acquired and lost motility, as sperm viability is lost, rendering the sperm useless for ICSI.
Typical criteria for poor sperm production, including an elevated serum FSH level or decreased testicular volume, does not predict which patients have sperm found with testicular sperm extraction (TESE). Even the histologic patterns on diagnostic testicular biopsy (Sertoli cell-only, maturation arrest, hypospermatogenesis) cannot perfectly predict the chance of finding sperm with TESE. However, many centers require diagnostic biopsies prior to TESE procedures to rule out carcinoma-in-situ (intratubular germ cell neoplasia) that is present in up to 3% of men with NOA who are candidates for treatment with TESE-ICSI.

